Unveiling the Partial Pressure Calculator, your ultimate tool for understanding and calculating partial pressures in gas mixtures. Dive into the world of gas dynamics and explore the significance of partial pressure in various scientific fields.
From understanding respiratory physiology to ensuring safety in industrial gas handling, partial pressure plays a crucial role. Let’s delve into the intricacies of partial pressure measurement techniques and their applications.
Partial Pressure Definitions and Principles
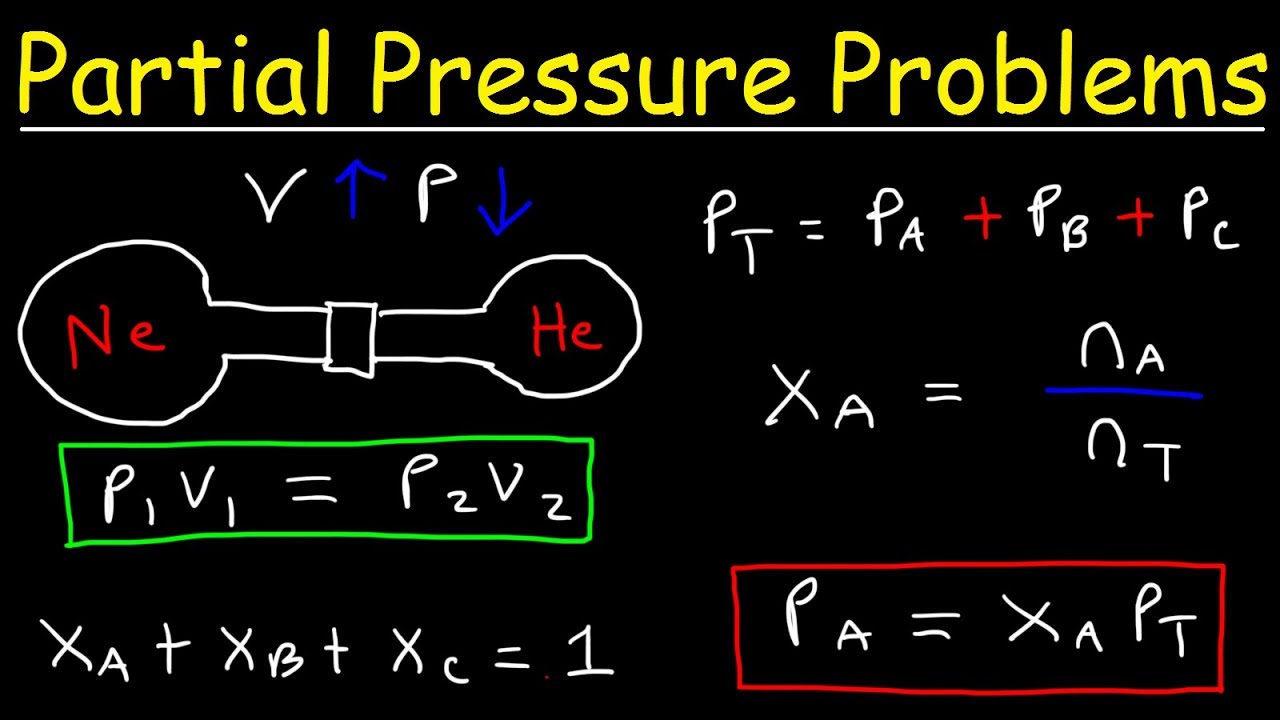
Partial pressure is a crucial concept in various scientific disciplines, particularly chemistry, physics, and physiology. It represents the pressure exerted by a specific gas in a mixture of gases. Understanding partial pressure is essential for comprehending the behavior of gases in diverse systems, such as the atmosphere, respiratory systems, and industrial processes.
Dalton’s Law and Partial Pressure
Dalton’s Law, also known as the Law of Partial Pressures, is a fundamental principle in gas mixture analysis. It states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas.
This law provides a convenient method for calculating the partial pressure of a specific gas within a mixture.
Measuring Partial Pressure
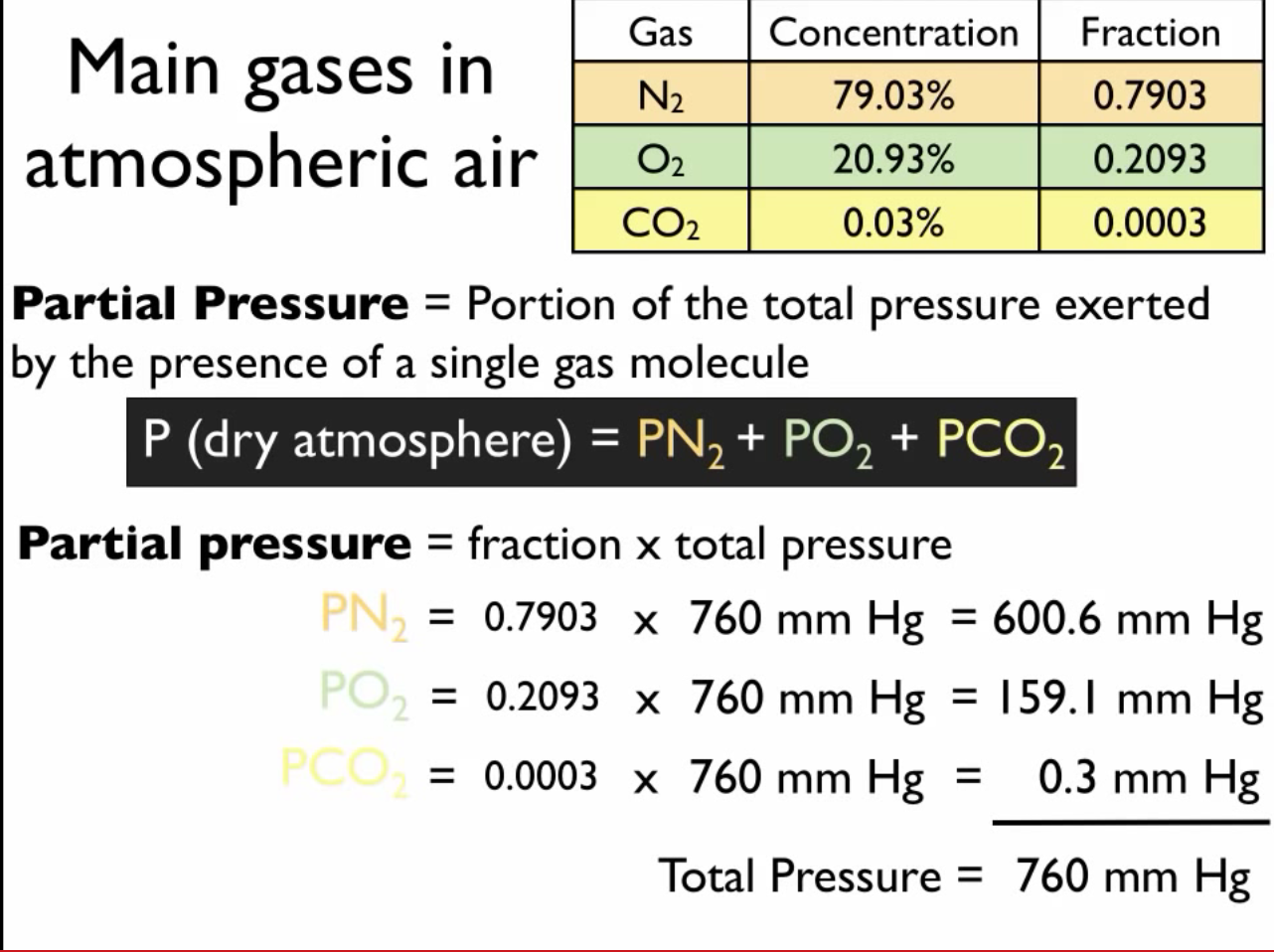
Partial pressure measurements are conducted in various applications. In meteorology, partial pressure is used to determine the concentration of gases in the atmosphere, such as oxygen, nitrogen, and carbon dioxide. In physiology, partial pressure is measured in blood samples to assess the respiratory function of the lungs.
In industrial settings, partial pressure measurements are crucial for optimizing chemical reactions and maintaining safe working conditions.
Partial Pressure Measurement Techniques: Partial Pressure Calculator

Measuring partial pressures accurately is crucial in various fields, including medicine, environmental monitoring, and industrial processes. Several techniques are available for this purpose, each with its advantages and limitations. Understanding these techniques is essential for selecting the most appropriate method based on specific requirements.
Manometry
Manometry is a simple and widely used technique for measuring partial pressures. It involves using a manometer, a device that measures pressure differences between two points. In this method, a gas sample is isolated in a chamber, and its pressure is compared to a reference pressure, typically atmospheric pressure.
The difference in pressure provides an indication of the partial pressure of the gas being measured.
Advantages:
- Simple and cost-effective
- Suitable for measuring relatively high partial pressures
Limitations:
- Not suitable for measuring very low partial pressures
- Can be affected by temperature and volume changes
Gas Chromatography
Gas chromatography (GC) is a versatile technique that can separate and quantify different gases in a sample. It involves passing a gas sample through a column packed with a stationary phase. Different gases interact with the stationary phase to varying degrees, causing them to elute at different times.
The partial pressure of each gas can be determined by measuring its retention time and comparing it to known standards.
Advantages:
- Can measure multiple gases simultaneously
- Suitable for measuring both high and low partial pressures
- Provides detailed information about the composition of the gas sample
Limitations:
- More expensive and complex than manometry
- May require sample preparation and calibration
Electrochemical Sensors, Partial pressure calculator
Electrochemical sensors are devices that convert a chemical signal into an electrical signal. In the context of partial pressure measurement, electrochemical sensors are typically used to measure the partial pressure of oxygen. These sensors rely on the principle of oxygen diffusion across a semi-permeable membrane.
The rate of oxygen diffusion is directly proportional to the partial pressure of oxygen in the gas sample.
Advantages:
- Compact and portable
- Suitable for continuous monitoring
- Can be used in a wide range of applications
Limitations:
- Not suitable for measuring very high partial pressures
- May require calibration and maintenance
Selecting the Appropriate Measurement Method
The choice of partial pressure measurement technique depends on the specific requirements of the application. Factors to consider include the following:
- Range of partial pressures to be measured
- Accuracy and precision required
- Availability of resources and expertise
- Cost and time constraints
By carefully considering these factors, it is possible to select the most appropriate partial pressure measurement technique for the intended purpose.
Partial Pressure Calculations in Gas Mixtures
Calculating partial pressures in gas mixtures involves determining the pressure exerted by each individual gas within the mixture. This is important in various fields, such as chemistry, environmental science, and medical applications. Understanding the principles and techniques of partial pressure calculations is crucial for accurate analysis and interpretation of gas mixtures.
The partial pressure of a gas is the pressure it would exert if it occupied the entire volume of the mixture alone. It is directly proportional to the mole fraction of the gas in the mixture. The mole fraction represents the ratio of the number of moles of the gas to the total number of moles of all gases in the mixture.
Mathematical Equations
The partial pressure of a gas can be calculated using the following equation:
Ppartial= P totalx X gas
where:
- P partialis the partial pressure of the gas
- P totalis the total pressure of the gas mixture
- X gasis the mole fraction of the gas in the mixture
Dalton’s Law of Partial Pressures
Dalton’s Law of Partial Pressures states that the total pressure exerted by a mixture of non-reacting gases is equal to the sum of the partial pressures of each individual gas. This law is valid for ideal gas mixtures, where the gases behave independently and do not interact with each other.
Applications of Partial Pressure Calculations
Partial pressure calculations have various applications, including:
- Determining the composition of gas mixtures
- Predicting the behavior of gases in chemical reactions
- Understanding gas exchange in biological systems
- Designing and optimizing gas separation processes
Partial Pressure Calculators
Partial pressure calculators are tools that simplify the calculation of partial pressures in gas mixtures. These calculators use mathematical equations and tables to determine the partial pressure of each gas based on the total pressure and mole fractions of the gases in the mixture.
Partial pressure calculators are widely used in research, industry, and educational settings.
Examples
Consider a gas mixture containing nitrogen (N 2), oxygen (O 2), and argon (Ar) with a total pressure of 1 atm. The mole fractions of N 2, O 2, and Ar are 0.7, 0.2, and 0.1, respectively.
Using the equation P partial= P totalx X gas, we can calculate the partial pressures of each gas:
- P N2= 1 atm x 0.7 = 0.7 atm
- P O2= 1 atm x 0.2 = 0.2 atm
- P Ar= 1 atm x 0.1 = 0.1 atm
Therefore, the partial pressures of N 2, O 2, and Ar in the mixture are 0.7 atm, 0.2 atm, and 0.1 atm, respectively.
Applications of Partial Pressure in Various Fields

Partial pressure finds applications in diverse fields, ranging from respiratory physiology and diving to industrial gas handling. It plays a crucial role in understanding gas exchange and respiratory function, designing diving equipment, and ensuring safety in industrial settings.
Respiratory Physiology
In respiratory physiology, partial pressure helps us comprehend the exchange of gases between the lungs and blood. The partial pressure of oxygen (pO2) and carbon dioxide (pCO2) in the lungs and blood determines the direction and rate of gas flow.
Understanding these partial pressures is essential for diagnosing and treating respiratory disorders, such as hypoxia and hypercapnia.
Diving
In diving, partial pressure is crucial for designing diving equipment and ensuring diver safety. The partial pressure of nitrogen in the breathing gas affects the risk of decompression sickness, a potentially life-threatening condition. Divers must carefully monitor the partial pressure of gases in their breathing mixtures to avoid exceeding safe limits.
Industrial Gas Handling
In industrial settings, partial pressure is essential for handling gases safely. The partial pressure of flammable gases, such as methane and propane, determines their flammability and explosion risk. By controlling the partial pressure of these gases, industries can minimize the risk of accidents and protect workers.
Closing Notes
Harnessing the power of partial pressure calculators, we’ve simplified the process of calculating partial pressures in gas mixtures. Whether you’re a researcher, a medical professional, or an industrial engineer, this tool empowers you with precise and efficient calculations.
