Welcome to the world of Boyle’s Law, where the Boyle’s Law calculator takes center stage, providing you with a powerful tool to explore the intricate relationship between pressure and volume in gases. Join us on an enlightening journey as we delve into the depths of this scientific marvel, unlocking its secrets and empowering you with a deeper understanding of gas behavior.
Our Boyle’s Law calculator is meticulously designed to cater to your scientific needs, offering a user-friendly interface and comprehensive features. Whether you’re a seasoned researcher, a curious student, or simply fascinated by the wonders of physics, this calculator is your indispensable companion, ready to assist you in unraveling the mysteries of gas dynamics.
Understanding Boyle’s Law Calculator
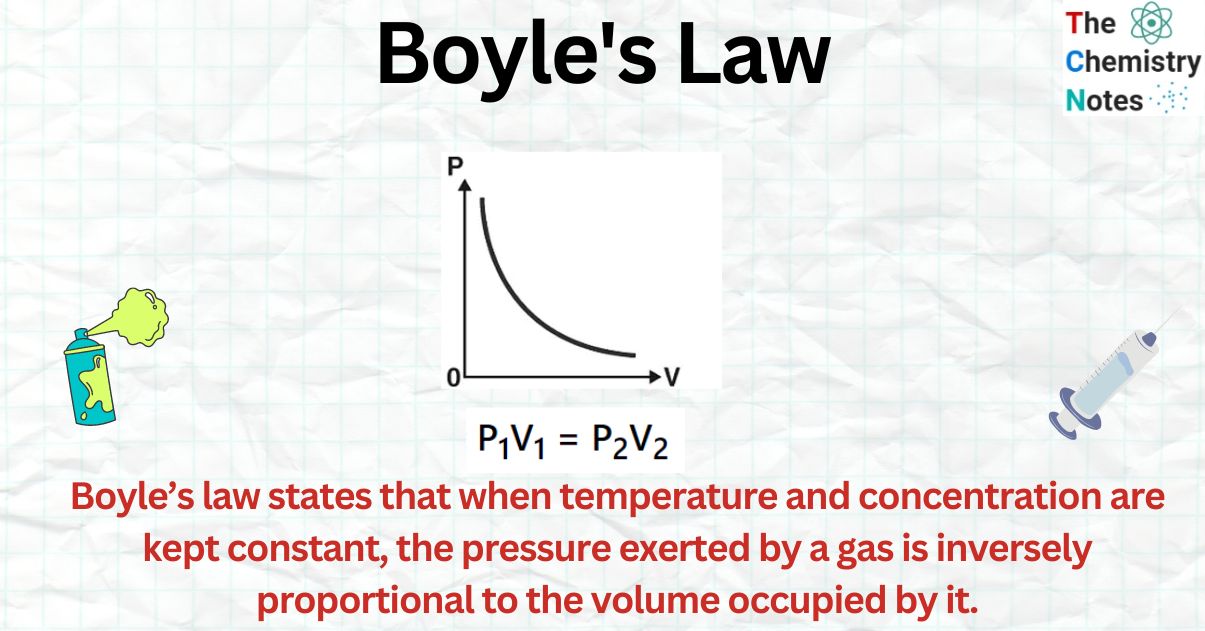
Boyle’s Law, also known as the Boyle-Mariotte Law, describes the inverse relationship between the pressure and volume of a gas at constant temperature. Mathematically, it can be expressed as: P₁V₁ = P₂V₂.
A Boyle’s Law calculator is an online tool designed to simplify the calculations involved in applying Boyle’s Law. It allows users to determine the unknown values (pressure or volume) when the other parameters are known.
Key Features and Parameters
- Pressure (P):The pressure of the gas, typically measured in atmospheres (atm), pascals (Pa), or pounds per square inch (psi).
- Volume (V):The volume of the gas, usually expressed in liters (L), cubic meters (m³), or milliliters (mL).
- Temperature (T):The temperature of the gas, which must remain constant for Boyle’s Law to apply. It is typically measured in Kelvin (K) or degrees Celsius (°C).
- Solve for:Users can select which parameter (pressure or volume) they want to solve for, given the known values of the other parameters.
Using the Calculator for Different Applications
The Boyle’s Law calculator finds applications in various scientific fields, from physics and chemistry to engineering and environmental science. Its versatility makes it a valuable tool for understanding and analyzing gas behavior.
Calculating Gas Properties, Boyle’s law calculator
The calculator allows you to determine the volume, pressure, or temperature of a gas when one or more of these variables changes. This is useful for predicting gas behavior in various scenarios, such as changes in altitude, temperature, or volume.
To use the calculator, simply input the known values and select the unknown variable you wish to calculate. The calculator will provide the result based on the Boyle’s Law equation.
Gas Law Calculations in Engineering
In engineering, the Boyle’s Law calculator aids in designing and analyzing systems involving gases. For instance, it helps determine the volume of a gas cylinder needed for a specific application or predict the pressure changes in a gas pipeline.
Applications in Environmental Science
Environmental scientists use the calculator to study the effects of changes in atmospheric pressure on gas behavior. It helps predict the impact of altitude or weather conditions on gas concentrations and pollution levels.
Advanced Features and Considerations
The Boyle’s Law calculator may offer additional features to enhance its functionality and usability.
It’s crucial to recognize the limitations and assumptions associated with using the calculator. This ensures accurate and efficient utilization.
Unit Conversions
The calculator may provide options for converting between different units of pressure and volume. This feature is helpful when dealing with experimental data or calculations that involve different units.
Graphical Representations
Some calculators offer graphical representations of the Boyle’s Law relationship. These graphs can provide a visual representation of the changes in pressure and volume, making it easier to understand the underlying concepts.
Tips for Accurate Use
- Input accurate values for pressure and volume.
- Ensure the units of measurement are consistent throughout the calculation.
- Understand the assumptions of Boyle’s Law and its limitations (e.g., ideal gas behavior).
Comparing Different Calculators
Selecting the most appropriate Boyle’s Law calculator for a specific task requires careful consideration of various factors, including features, accuracy, and ease of use. To facilitate this decision-making process, we have compiled a comparative analysis of different calculators, highlighting their strengths, weaknesses, and recommended applications.
-
Features
Different calculators offer varying sets of features. Some may provide basic calculations, while others include advanced capabilities such as handling multiple gases, temperature conversions, and graphing. Consider the specific features required for your task to identify the most suitable calculator.
-
Accuracy
The accuracy of a calculator is crucial for reliable results. Look for calculators that employ well-established equations and algorithms, and consider user reviews or independent evaluations to assess their accuracy.
-
Ease of Use
A user-friendly interface is essential for efficient calculations. Choose calculators with clear instructions, intuitive navigation, and minimal input requirements. If you need to perform complex calculations, consider calculators that offer step-by-step guidance or allow customization of input parameters.
By evaluating these factors and comparing different calculators, you can select the most appropriate tool for your Boyle’s Law calculations, ensuring accurate and efficient results.
Final Summary: Boyle’s Law Calculator

As we bid farewell to our exploration of Boyle’s Law, we hope that this calculator has ignited a spark of scientific curiosity within you. Remember, the pursuit of knowledge is an ongoing adventure, and we encourage you to continue your exploration into the fascinating realm of physics.
Let this calculator be your trusted guide, empowering you to unravel the complexities of the natural world with precision and ease.
