Embark on a journey into the fascinating world of thermodynamics with our specific heat capacity calculator. This tool empowers you to delve into the intricate details of thermal properties, unlocking a deeper understanding of how substances respond to temperature changes.
As you navigate this comprehensive guide, you’ll uncover the secrets of specific heat capacity, its influential factors, and its practical applications in diverse fields. Prepare to unravel the mysteries of thermal behavior and harness the power of this essential concept.
Specific Heat Capacity Calculation: Specific Heat Capacity Calculator
Specific heat capacity is a physical property of matter that describes the amount of heat energy required to raise the temperature of one gram of a substance by one degree Celsius.
The formula for calculating specific heat capacity is:
$C = Q / (m
ΔT)$
Where:
- $C$ is the specific heat capacity in J/g°C
- $Q$ is the amount of heat energy in joules
- $m$ is the mass of the substance in grams
- $ΔT$ is the change in temperature in °C
Examples of Specific Heat Capacity Calculations
For example, if you add 100 joules of heat to 10 grams of water and the temperature increases by 2°C, the specific heat capacity of water would be:
$C = 100 J / (10 g
2°C) = 5 J/g°C$
Units of Measurement for Specific Heat Capacity, Specific heat capacity calculator
The SI unit of specific heat capacity is joules per gram per degree Celsius (J/g°C). Other commonly used units include calories per gram per degree Celsius (cal/g°C) and British thermal units per pound per degree Fahrenheit (Btu/lb°F).
Factors Affecting Specific Heat Capacity
The specific heat capacity of a substance is not a fixed property; it can vary depending on several factors, including its molecular structure and temperature.
Molecular Structure
The molecular structure of a substance significantly influences its specific heat capacity. Substances with simpler molecular structures, such as monatomic gases like helium, tend to have lower specific heat capacities compared to substances with complex molecular structures, such as organic compounds.
The reason for this difference is that more complex molecules have more degrees of freedom, meaning they can store energy in various ways. For example, in addition to translational and rotational motion, organic molecules can also vibrate and undergo conformational changes.
These additional modes of energy storage contribute to the higher specific heat capacity of complex molecules.
Temperature
The specific heat capacity of a substance can also vary with temperature. In general, the specific heat capacity of a substance increases with increasing temperature. This is because, at higher temperatures, molecules have more energy and can store more energy in various ways.
However, there are exceptions to this rule. For example, the specific heat capacity of water decreases slightly with increasing temperature above 35 °C.
Applications of Specific Heat Capacity
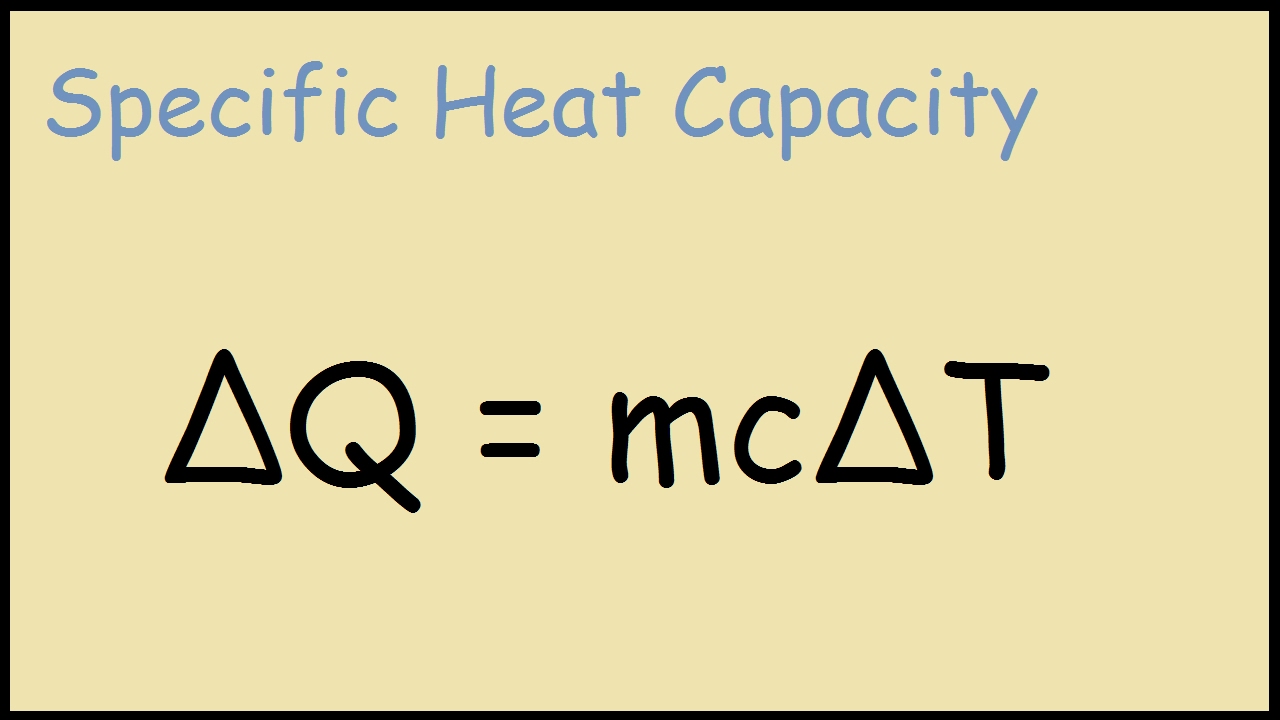
Specific heat capacity finds extensive applications in various scientific and engineering disciplines, including engineering, chemistry, and physics. It plays a crucial role in the design and optimization of thermal systems, allowing engineers and scientists to predict and control heat transfer and temperature changes in different materials and systems.
Engineering
In engineering, specific heat capacity is essential for designing thermal systems such as heat exchangers, boilers, and cooling systems. It helps engineers determine the amount of heat required to raise the temperature of a substance or the amount of heat released when a substance cools.
This information is critical for optimizing system performance, ensuring efficient heat transfer, and preventing overheating or undercooling.
- HVAC systems:Specific heat capacity is used to calculate the heating and cooling loads of buildings, ensuring comfortable indoor temperatures and energy efficiency.
- Power plants:Engineers use specific heat capacity to design boilers and condensers, optimizing heat transfer and maximizing energy production.
- Automotive engineering:Specific heat capacity is considered in the design of engines, cooling systems, and exhaust systems to manage heat dissipation and prevent overheating.
Chemistry
In chemistry, specific heat capacity is used to determine the heat absorbed or released during chemical reactions and phase changes. It helps chemists understand the thermodynamics of reactions and predict the temperature changes associated with chemical processes.
- Calorimetry:Specific heat capacity is used in calorimetry experiments to measure the heat released or absorbed during chemical reactions, providing insights into reaction enthalpies.
- Phase transitions:Specific heat capacity is essential for calculating the heat required for melting, freezing, vaporization, and condensation, enabling chemists to study phase behavior and energy changes.
- Thermochemistry:Specific heat capacity is used to calculate the heat capacity of solutions and mixtures, aiding in the understanding of solution thermodynamics and equilibrium.
Physics
In physics, specific heat capacity is used to study heat transfer, thermal conductivity, and thermodynamics. It helps physicists understand the behavior of materials under different temperature conditions and predict heat flow in various systems.
- Heat transfer:Specific heat capacity is used to calculate the heat flow rate in materials and systems, enabling the design of efficient heat transfer devices and systems.
- Thermal conductivity:Specific heat capacity is related to thermal conductivity, allowing physicists to study the ability of materials to conduct heat.
- Thermodynamics:Specific heat capacity is used in thermodynamic equations to calculate entropy changes, heat capacities, and other thermodynamic properties of materials.
Comparison of Specific Heat Capacities

Specific heat capacity is a measure of the amount of heat required to raise the temperature of a unit mass of a substance by one degree Celsius or one Kelvin. Different materials have different specific heat capacities, which means that they require different amounts of heat to raise their temperatures by the same amount.
The table below compares the specific heat capacities of some common materials:
| Material | Specific Heat Capacity (J/g°C) |
|---|---|
| Water | 4.187 |
| Ice | 2.09 |
| Aluminum | 0.903 |
| Iron | 0.452 |
| Copper | 0.385 |
| Glass | 0.84 |
| Wood | 1.76 |
As can be seen from the table, water has the highest specific heat capacity of all the materials listed. This means that it takes more heat to raise the temperature of water by one degree Celsius than it does to raise the temperature of any of the other materials by the same amount.
The high specific heat capacity of water makes it a good material for use in thermal applications, such as heating and cooling systems. Water can absorb or release a large amount of heat without changing its temperature significantly, which makes it an effective medium for transferring heat.
Materials with low specific heat capacities, such as metals, are good conductors of heat. This means that they can transfer heat quickly and efficiently. Metals are often used in applications where heat transfer is important, such as in cookware and heat exchangers.
The specific heat capacity of a material is an important property to consider when selecting materials for thermal applications. By understanding the specific heat capacities of different materials, engineers can design systems that are efficient and effective.
Wrap-Up
Through this exploration of specific heat capacity, we’ve illuminated the intricate relationship between temperature and thermal energy. From the depths of molecular structure to the design of efficient thermal systems, specific heat capacity stands as a cornerstone of thermodynamics. May this newfound knowledge empower you to conquer thermal challenges and unlock the full potential of your designs.
