Introducing the net ionic calculator, an indispensable tool for delving into the intricate world of chemical reactions. This interactive instrument empowers you to unravel the complexities of net ionic equations, providing a deeper understanding of the fundamental principles that govern chemical transformations.
Beyond mere calculations, the net ionic calculator serves as a gateway to unraveling the hidden mechanisms behind chemical reactions. It unveils the true essence of chemical interactions, enabling you to decipher the symphony of ions and molecules as they dance through intricate transformations.
Net Ionic Equations

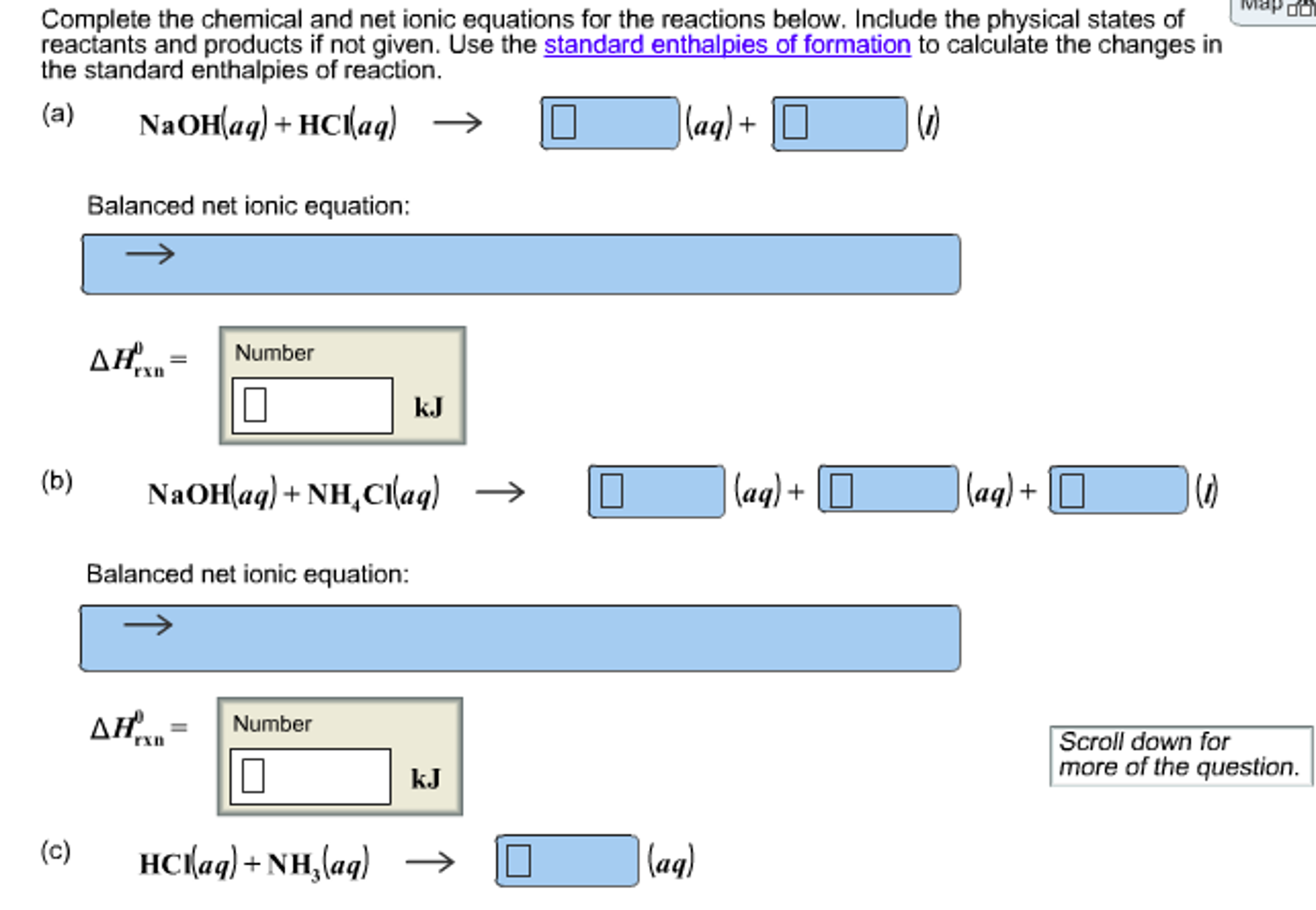
In chemistry, a net ionic equation is a chemical equation that shows only the ions that are actually reacting in a chemical reaction. The ions that do not participate in the reaction are called spectator ions and are omitted from the net ionic equation.
Net ionic equations are useful for understanding the chemical reactions that occur in aqueous solutions. In an aqueous solution, water molecules are present and can participate in the reaction. The water molecules can act as a solvent, a reactant, or a product.
The spectator ions are the ions that do not participate in the reaction and are present in the solution before and after the reaction.
Molecular Equations
A molecular equation is a chemical equation that shows all of the reactants and products of a chemical reaction. The molecular equation includes the chemical formulas of the reactants and products, as well as the coefficients that balance the equation.
Difference Between Molecular Equations and Net Ionic Equations
The main difference between a molecular equation and a net ionic equation is that the net ionic equation only shows the ions that are actually reacting in the chemical reaction. The spectator ions are omitted from the net ionic equation.
For example, the molecular equation for the reaction between sodium chloride and silver nitrate is:
NaCl + AgNO3→ AgCl + NaNO 3
The net ionic equation for this reaction is:
Na++ Cl –+ Ag ++ NO 3–→ AgCl + Na ++ NO 3–
The spectator ions in this reaction are sodium and nitrate. These ions do not participate in the reaction and are present in the solution before and after the reaction.
Net Ionic Calculator
A net ionic calculator is a tool that can be used to determine the net ionic equation for a given chemical reaction. Net ionic equations show only the ions that are actually reacting in a solution, and they can be used to predict the products of a reaction and to calculate the equilibrium constant.
To use a net ionic calculator, you will need to enter the balanced chemical equation for the reaction. The calculator will then use this information to determine the net ionic equation. Here is a step-by-step guide on how to use a net ionic calculator:
Step 1: Enter the balanced chemical equation
The first step is to enter the balanced chemical equation for the reaction. The balanced chemical equation shows the chemical formulas of the reactants and products, as well as the coefficients that balance the equation. For example, the balanced chemical equation for the reaction between sodium chloride and silver nitrate is:
“`NaCl + AgNO3
> AgCl + NaNO3
“`
Step 2: Select the ions that are reacting
Once you have entered the balanced chemical equation, you will need to select the ions that are actually reacting. In the example above, the ions that are reacting are Na+ and Cl- from NaCl, and Ag+ and NO3- from AgNO3.
To select the ions, you will need to click on the “Select Ions” button. The calculator will then display a list of the ions that are present in the reaction. You will need to select the ions that are reacting by clicking on the checkboxes next to their names.
Step 3: Click the “Calculate” button
Once you have selected the ions that are reacting, you will need to click on the “Calculate” button. The calculator will then use this information to determine the net ionic equation. The net ionic equation will be displayed in the “Net Ionic Equation” field.
Advantages of using a net ionic calculator
- Net ionic calculators are easy to use.
- Net ionic calculators can save you time.
- Net ionic calculators can help you to understand chemical reactions.
Disadvantages of using a net ionic calculator
- Net ionic calculators can be inaccurate if the balanced chemical equation is not entered correctly.
- Net ionic calculators cannot be used to predict the products of a reaction if the reaction is not a simple acid-base reaction or precipitation reaction.
Applications of Net Ionic Equations
Net ionic equations are powerful tools used in chemistry to simplify and understand complex reactions. They provide a clear representation of the chemical changes that occur during a reaction, allowing us to focus on the essential aspects of the process.
Applications in Real-World Scenarios, Net ionic calculator
Net ionic equations have numerous applications in real-world scenarios. For example, they are used in:
- Predicting the products of a reaction:By identifying the ions that participate in a reaction, we can use net ionic equations to predict the products that will form.
- Determining the solubility of compounds:Net ionic equations can help us understand the factors that affect the solubility of compounds. By analyzing the ions involved in a precipitation reaction, we can determine whether a precipitate will form or not.
- Balancing chemical equations:Net ionic equations can be used to balance chemical equations in a more efficient and straightforward manner.
Importance in Understanding Chemical Reactions
Net ionic equations are crucial for understanding chemical reactions because they:
- Eliminate spectator ions:Spectator ions are ions that do not participate in the chemical change and are present on both sides of the equation. Net ionic equations remove these ions, allowing us to focus on the essential ions that undergo the reaction.
- Highlight the essential changes:By isolating the ions that participate in the reaction, net ionic equations make it easier to identify the changes that occur during the reaction.
- Provide a simplified representation:Net ionic equations simplify complex reactions, making them easier to understand and interpret.
Advanced Topics in Net Ionic Equations
Net ionic equations provide a simplified representation of chemical reactions by focusing on the ions that participate in the reaction. Advanced topics in net ionic equations include redox reactions, electrochemistry, and limitations of net ionic equations.
Redox Reactions
Redox reactions involve the transfer of electrons between atoms or ions. In net ionic equations, redox reactions can be represented by identifying the species that are oxidized (lose electrons) and reduced (gain electrons). The half-reactions for oxidation and reduction are then combined to form the overall net ionic equation.
Electrochemistry
Electrochemistry deals with the relationship between electrical energy and chemical reactions. Net ionic equations can be used to represent electrochemical reactions, such as those occurring in batteries or electrolysis cells. In these reactions, the net ionic equation shows the ions that are involved in the transfer of electrons at the electrodes.
Limitations of Net Ionic Equations
Net ionic equations have certain limitations. They do not provide information about:
- The physical state of the reactants and products (e.g., solid, liquid, gas)
- The concentration of the reactants and products
- The rate of the reaction
- The equilibrium constant of the reaction
Outcome Summary

In conclusion, the net ionic calculator stands as a formidable ally for chemists and students alike, offering a profound understanding of chemical reactions. Its ability to simplify complex equations and reveal the underlying ionic interactions makes it an invaluable asset in the pursuit of chemical knowledge.
Embrace the power of this tool, and unlock the secrets of chemical reactions with newfound clarity and precision.
