Coulombs law calculator – Embark on an electrifying journey with our Coulomb’s Law Calculator, a powerful tool that unlocks the mysteries of electrostatics. Dive into the world of charged particles and their interactions, where Coulomb’s Law reigns supreme.
This comprehensive guide will illuminate the intricacies of Coulomb’s Law, its practical applications, and the nuances of using our calculator. Get ready to harness the power of electrostatics and explore the fascinating world of charged particles.
Coulomb’s Law Calculator Overview: Coulombs Law Calculator

Coulomb’s law calculator is an online tool designed to assist users in calculating the electrostatic force between two charged particles. It utilizes Coulomb’s law, a fundamental principle in electrostatics, to determine the magnitude and direction of the force based on the charges and distance between the particles.
Coulomb’s law is crucial in understanding and analyzing electrostatic interactions, which play a significant role in various scientific and engineering fields, including physics, chemistry, and electrical engineering. By providing accurate calculations based on Coulomb’s law, the calculator serves as a valuable resource for students, researchers, and practitioners.
Coulomb’s Law
Coulomb’s law states that the electrostatic force ( F) between two point charges ( q1and q2) separated by a distance ( r) is directly proportional to the product of the charges and inversely proportional to the square of the distance between them.
Mathematically, it can be expressed as:
F = k
- (q1
- q 2) / r 2
where kis Coulomb’s constant, approximately 8.988 × 10 9N m 2/C 2.
Using a Coulomb’s Law Calculator
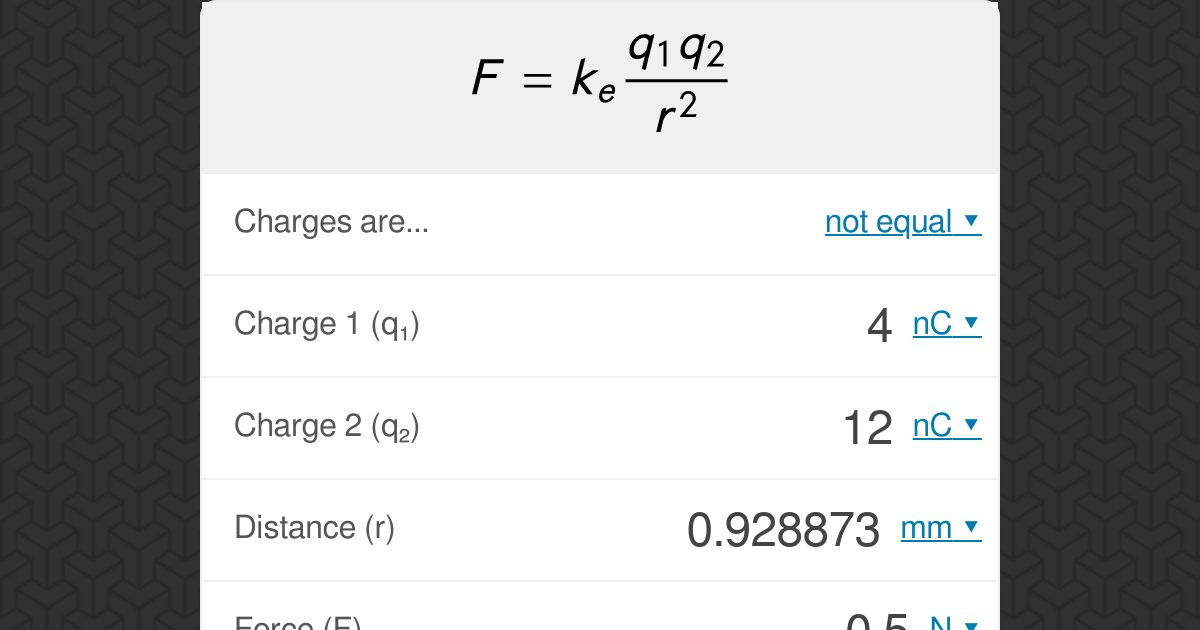
Using a Coulomb’s law calculator is a convenient way to determine the electrostatic force between two charged objects. Here’s a step-by-step guide on how to use one:
- Enter the charges of the objects:Input the values of the charges in coulombs (C) for both objects. If the charges are equal, enter the same value for both.
- Specify the distance between the objects:Enter the distance between the centers of the objects in meters (m).
- Select the units for the result:Choose the desired units for the calculated force, such as newtons (N) or dynes (dyn).
- Calculate the force:Click the “Calculate” button to obtain the electrostatic force between the objects.
Here’s an example table with labeled columns for input values and calculated results:
| Input | Value |
|---|---|
| Charge of object 1 (Q1) | 2 μC (2 × 10-6 C) |
| Charge of object 2 (Q2) | -3 μC (-3 × 10-6 C) |
| Distance between objects (r) | 0.5 m |
| Calculated Force | 14.4 N |
The calculator takes into account the Coulomb’s law formula, F = k – (Q1 – Q2) / r 2, where:
- F is the electrostatic force in newtons (N)
- k is Coulomb’s constant, approximately 8.98755 × 10 9N⋅m 2/C 2
- Q1 and Q2 are the charges of the objects in coulombs (C)
- r is the distance between the objects in meters (m)
The calculator automatically converts the input values to the appropriate units to ensure accurate results. For example, if the charges are entered in microcoulombs (μC), the calculator will convert them to coulombs (C) before performing the calculation.
Applications of Coulomb’s Law
.png)
Coulomb’s law finds practical applications in various scientific disciplines, including engineering, physics, and chemistry. It provides a fundamental understanding of electrostatic interactions and enables the calculation of forces between charged particles.
In engineering, Coulomb’s law is used in the design of electrical circuits, capacitors, and other electronic components. It helps determine the distribution of electric fields and the forces acting on charged particles within these systems.
In Physics
- Coulomb’s law is essential in understanding the behavior of charged particles in particle accelerators and plasma physics.
- It plays a crucial role in the study of electromagnetism, helping physicists analyze the forces between charged objects and the behavior of electric fields.
In Chemistry
- Coulomb’s law is used to calculate the electrostatic forces between ions in ionic compounds, providing insights into their stability and properties.
- It aids in understanding the behavior of electrolytes in solutions and the formation of ionic bonds.
Limitations and Considerations

While Coulomb’s law calculators provide a convenient and efficient way to estimate electrostatic forces, it’s essential to be aware of their limitations and the factors that can affect the accuracy of the calculations.
One limitation of Coulomb’s law calculators is that they assume a point-like distribution of charges. In reality, charges are often distributed over a finite volume, which can lead to inaccuracies in the calculated force. Additionally, the calculations assume that the charges are stationary, which may not always be the case in practical applications.
Accuracy Factors
Several factors can affect the accuracy of Coulomb’s law calculations:
- Distance between charges:The accuracy of the calculation decreases as the distance between the charges increases. This is because the electrostatic force is inversely proportional to the square of the distance.
- Presence of other charges:The presence of other charges in the vicinity can affect the electrostatic force between the two charges of interest. This is because the electrostatic force is a vector quantity, and the forces due to different charges can add or cancel each other out.
- Dielectric constant of the medium:The dielectric constant of the medium between the charges can affect the strength of the electrostatic force. A higher dielectric constant reduces the force between the charges.
Interpreting Results, Coulombs law calculator
When interpreting the results from a Coulomb’s law calculator, it’s important to consider the limitations and accuracy factors discussed above. The calculated force may not be exact, but it can provide a reasonable estimate for many applications.
It’s also important to remember that Coulomb’s law only describes the electrostatic force between point charges. In practical applications, charges are often distributed over a finite volume, and the electrostatic force may be more complex than the simple Coulombic interaction.
Conclusion
As we conclude our exploration of Coulomb’s Law, remember that this calculator is your gateway to understanding the fundamental principles of electrostatics. Whether you’re an aspiring engineer, a curious physicist, or simply fascinated by the interactions of charged particles, this tool empowers you to delve deeper into the wonders of the electrostatic realm.
