Introducing the freezing point depression calculator, a powerful tool that empowers scientists and researchers to delve into the fascinating realm of freezing point depression. This remarkable tool unveils the secrets behind the freezing behavior of solutions, providing valuable insights into their composition and properties.
By harnessing the principles of freezing point depression, this calculator unlocks a treasure trove of applications in diverse fields, ranging from chemistry and biology to engineering. Prepare to embark on an enthralling journey as we explore the intricacies of freezing point depression and its captivating applications.
Calculations and Formula

Freezing point depression is a colligative property that describes the decrease in the freezing point of a solvent when a solute is added to it. The freezing point depression is directly proportional to the molality of the solution, which is the number of moles of solute per kilogram of solvent.
The formula for calculating freezing point depression is:
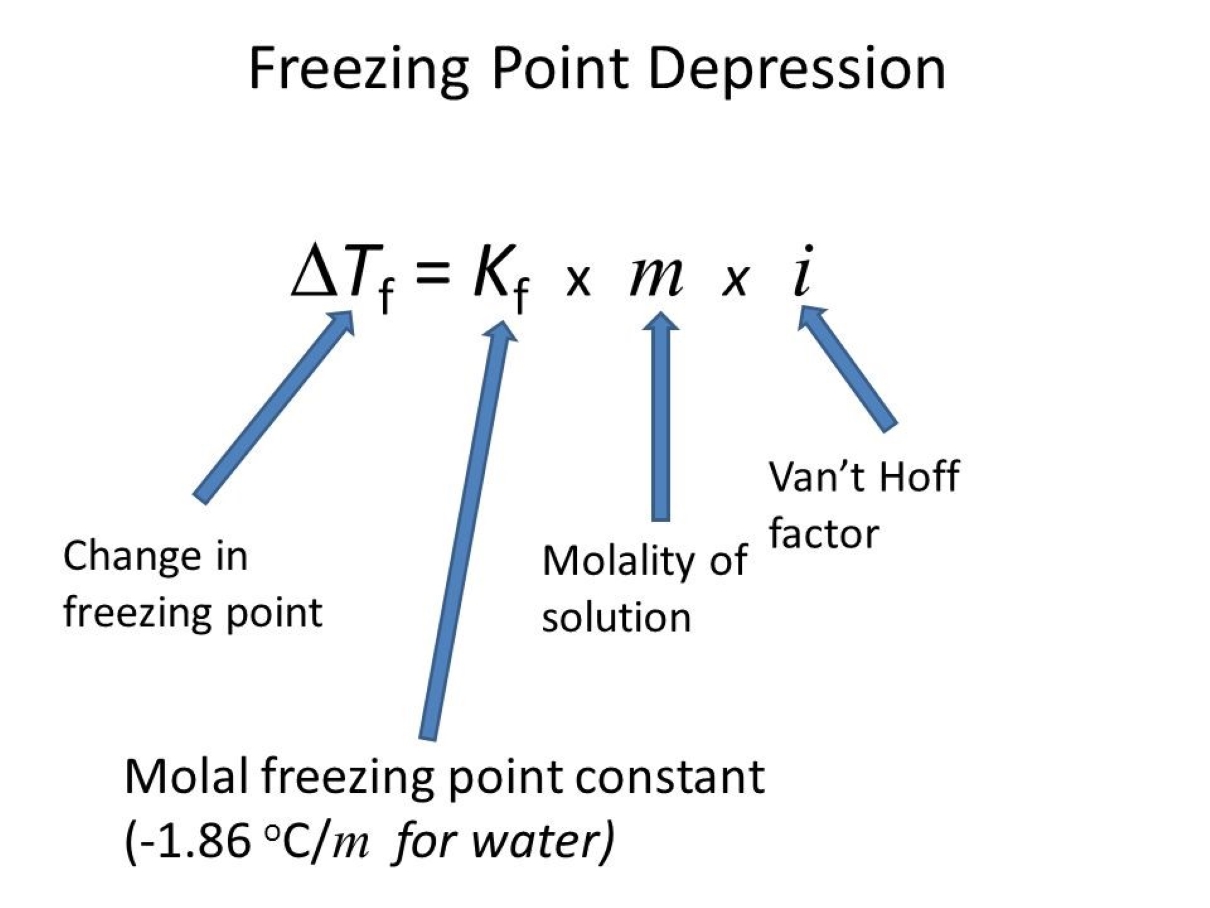
ΔTf = Kf
m
Where:
- ΔTf is the freezing point depression in degrees Celsius
- Kf is the cryoscopic constant of the solvent in degrees Celsius per molality
- m is the molality of the solution in molality
The following table shows how to use the formula to calculate the freezing point depression of a solution of NaCl in water:
| Solute | Molality (m) | Cryoscopic constant (Kf) | Freezing point depression (ΔTf) |
|---|---|---|---|
| NaCl | 0.1 | 1.86 | 0.186 |
| NaCl | 0.2 | 1.86 | 0.372 |
| NaCl | 0.3 | 1.86 | 0.558 |
Factors Affecting Freezing Point Depression
Freezing point depression is influenced by several factors, including the concentration of the solute, the properties of the solvent, and the temperature of the solution.
Solute Concentration
The freezing point depression of a solution is directly proportional to the concentration of the solute. As the concentration of the solute increases, the freezing point of the solution decreases. This is because the solute particles interfere with the formation of ice crystals, making it more difficult for the solvent molecules to freeze.
For example, a solution of 1 mol/kg of NaCl has a freezing point of -3.4 °C, while a solution of 2 mol/kg of NaCl has a freezing point of -6.8 °C.
Solvent Properties
The freezing point depression of a solution is also affected by the properties of the solvent. Solvents with a higher freezing point have a greater ability to interfere with the formation of ice crystals, resulting in a greater freezing point depression.
This is because the solvent molecules compete with the solute particles for space in the ice lattice.
For example, water has a freezing point of 0 °C, while methanol has a freezing point of -97.6 °C. A solution of 1 mol/kg of NaCl in water has a freezing point of -3.4 °C, while a solution of 1 mol/kg of NaCl in methanol has a freezing point of -2.4 °C.
Temperature
The freezing point depression of a solution is also affected by the temperature of the solution. As the temperature of the solution increases, the freezing point depression decreases. This is because the higher temperature provides more energy to the solvent molecules, making it easier for them to overcome the interference of the solute particles and form ice crystals.
For example, a solution of 1 mol/kg of NaCl has a freezing point of -3.4 °C at 0 °C, but a freezing point of -3.2 °C at 10 °C.
| Factor | Effect on Freezing Point Depression |
|---|---|
| Solute Concentration | Directly proportional |
| Solvent Properties | Greater freezing point depression for solvents with a higher freezing point |
| Temperature | Inversely proportional |
Applications of Freezing Point Depression
Freezing point depression finds numerous practical applications in various scientific fields. It serves as a valuable tool for determining solute concentration, purity, and other properties of solutions.
Chemistry
In chemistry, freezing point depression is used to determine the molecular weight of solutes. By measuring the freezing point depression of a solution, chemists can calculate the number of solute particles present in the solution. This information can then be used to determine the molecular weight of the solute.
Freezing point depression is also used to determine the purity of substances. By measuring the freezing point depression of a substance, chemists can determine the amount of impurities present in the substance. This information can then be used to assess the purity of the substance.
Biology
In biology, freezing point depression is used to determine the osmotic pressure of solutions. Osmotic pressure is the pressure that must be applied to a solution to prevent the passage of water across a semipermeable membrane. By measuring the freezing point depression of a solution, biologists can calculate the osmotic pressure of the solution.
Freezing point depression is also used to determine the water potential of plant cells. Water potential is the potential energy of water in a plant cell. By measuring the freezing point depression of a plant cell, biologists can calculate the water potential of the cell.
Engineering
In engineering, freezing point depression is used to design antifreeze solutions. Antifreeze solutions are used to prevent water from freezing in engines and other mechanical systems. By measuring the freezing point depression of an antifreeze solution, engineers can determine the concentration of antifreeze needed to prevent freezing at a desired temperature.
Freezing point depression is also used to design deicing solutions. Deicing solutions are used to remove ice from roads and other surfaces. By measuring the freezing point depression of a deicing solution, engineers can determine the concentration of deicing agent needed to effectively remove ice.
Advantages and Limitations
Freezing point depression is a simple and inexpensive technique that can be used to determine the solute concentration, purity, and other properties of solutions. However, there are some limitations to the use of freezing point depression.
One limitation of freezing point depression is that it can only be used to measure the concentration of nonvolatile solutes. Volatile solutes will evaporate during the freezing point depression experiment, which will lead to inaccurate results.
Another limitation of freezing point depression is that it is not very sensitive. Small changes in solute concentration will result in small changes in freezing point depression. This can make it difficult to accurately determine the concentration of solutes in very dilute solutions.
Despite these limitations, freezing point depression remains a valuable tool for determining the solute concentration, purity, and other properties of solutions.
Limitations and Considerations

Freezing point depression, while a useful technique, has certain limitations and considerations that must be acknowledged to ensure accurate and reliable results. These include the influence of impurities, non-ideal behavior of solutions, and potential experimental errors.
Impurities
The presence of impurities in the solvent or solute can significantly affect the observed freezing point depression. Impurities can act as nucleation sites, promoting crystallization and leading to an earlier freezing point. To minimize this effect, it is essential to use high-purity solvents and solutes.
Non-Ideal Behavior
The freezing point depression equation assumes ideal behavior of the solution, where solute particles do not interact with each other. However, in reality, solute particles can interact, leading to deviations from ideal behavior. These deviations can result in inaccuracies in the calculated molar mass or other properties.
Experimental Errors, Freezing point depression calculator
Experimental errors can also impact the accuracy of freezing point depression measurements. Errors can arise from inaccurate temperature measurements, incomplete dissolution of the solute, or contamination of the sample. Careful experimental technique and proper calibration of equipment are crucial to minimize these errors.
End of Discussion: Freezing Point Depression Calculator
As we conclude our exploration of freezing point depression, it is evident that this phenomenon holds immense significance in various scientific disciplines. The freezing point depression calculator serves as an invaluable tool, enabling researchers to unravel the mysteries of solutions and their components.
From determining solute concentrations to assessing purity levels, freezing point depression finds widespread use in both research and industrial settings. As we continue to push the boundaries of scientific understanding, this remarkable tool will undoubtedly play an increasingly pivotal role in shaping our knowledge of the molecular world.
