Understanding how to calculate percent recovery is essential for ensuring the accuracy and precision of analytical methods in various fields. This comprehensive guide will provide you with the formula, methods, applications, and troubleshooting tips to master this crucial aspect of chemical analysis.
Defining Percent Recovery
Percent recovery is a crucial concept in various scientific disciplines, including chemistry, biochemistry, and environmental science. It measures the efficiency of a process or procedure by comparing the amount of a substance recovered to the amount initially present.
In essence, percent recovery quantifies the proportion of a target analyte that is successfully extracted, purified, or otherwise recovered from a sample. It provides valuable insights into the effectiveness of experimental methods, allowing researchers and analysts to assess the accuracy and reliability of their results.
Significance of Percent Recovery
Percent recovery plays a pivotal role in evaluating the performance of analytical techniques, ensuring the validity and reliability of experimental data. It helps researchers:
- Identify and troubleshoot inefficiencies in extraction or purification procedures.
- Optimize experimental conditions to maximize recovery and minimize losses.
- Compare the effectiveness of different analytical methods or protocols.
- Ensure the accuracy and precision of quantitative measurements.
- Comply with regulatory standards and quality control requirements.
Formula and Calculation Methods

Calculating percent recovery involves utilizing a formula and employing various methods to determine the percentage of recovered analyte compared to the original amount.
Formula for Percent Recovery, How to calculate percent recovery
The formula for calculating percent recovery is:
Percent Recovery = (Amount Recovered / Amount Spiked) x 100
Where:
- Amount Recovered is the quantity of analyte measured in the sample after the extraction and analysis process.
- Amount Spiked is the known amount of analyte added to the sample before extraction.
Methods for Determining Recovery Percentage
There are several methods for determining recovery percentage, including:
- Internal Standard Method:Utilizes an internal standard, a compound similar to the analyte but not present in the sample, to compensate for variations in extraction and analysis.
- Standard Addition Method:Involves adding known amounts of analyte to the sample and measuring the response to determine the recovery efficiency.
- Surrogate Spike Method:Employs a surrogate, a compound chemically similar to the analyte but not expected to be present in the sample, to monitor extraction efficiency.
Recovery Factor and Yield
Recovery factor and yield are related concepts in percent recovery calculations:
- Recovery Factor:Represents the efficiency of the extraction and analysis process and is calculated by dividing the amount recovered by the amount spiked.
- Yield:Indicates the overall efficiency of the extraction and analysis process, considering both recovery and losses, and is calculated by multiplying the percent recovery by the extraction efficiency.
Applications in Chemical Analysis

Percent recovery plays a vital role in analytical chemistry, serving as a key indicator of the accuracy and precision of analytical methods. It helps assess the extent to which an analytical technique can extract and quantify the target analyte from a sample.
By comparing the measured concentration to the known or expected concentration, percent recovery provides valuable insights into the efficiency of the extraction, separation, and detection processes involved in the analysis.
Specific Analytical Techniques
Percent recovery is particularly crucial in various analytical techniques, including:
- Chromatography:In techniques like HPLC and GC, percent recovery helps evaluate the efficiency of the separation and detection processes, ensuring accurate quantification of target analytes.
- Spectrophotometry:Percent recovery is used to assess the accuracy of spectrophotometric measurements, such as in UV-Vis and fluorescence spectroscopy, by comparing the measured absorbance or emission intensity to the expected values.
- Electrochemical Analysis:In techniques like voltammetry and amperometry, percent recovery provides insights into the efficiency of electrode reactions and the sensitivity of the detection system.
- Immunoassays:Percent recovery is essential in immunoassays, such as ELISA and Western blotting, to evaluate the accuracy and specificity of antibody-antigen interactions.
Troubleshooting and Optimization: How To Calculate Percent Recovery
Ensuring accurate percent recovery values requires attention to various factors that can influence the results. By understanding these factors and implementing appropriate strategies, you can troubleshoot issues and optimize recovery.
Identifying Factors Affecting Percent Recovery
- Sample Preparation:Incomplete or improper sample preparation can lead to analyte loss or contamination, affecting recovery.
- Extraction Efficiency:The efficiency of the extraction method used can impact the amount of analyte extracted from the sample, influencing recovery.
- Matrix Effects:The presence of other compounds in the sample matrix can interfere with analyte extraction or detection, affecting recovery.
- Instrument Calibration:Accurate calibration of analytical instruments is crucial to ensure reliable recovery measurements.
- Analyst Variability:Differences in technique or interpretation among analysts can contribute to variability in recovery results.
Strategies for Troubleshooting and Optimization
To troubleshoot and optimize percent recovery, consider the following strategies:
- Review Sample Preparation:Ensure that the sample preparation method is appropriate for the analyte and matrix, and that it is performed carefully to minimize analyte loss or contamination.
- Optimize Extraction:Select an extraction method that efficiently recovers the analyte from the sample matrix. Consider using internal standards to monitor extraction efficiency.
- Address Matrix Effects:If matrix effects are suspected, employ sample cleanup techniques to remove interfering compounds or use matrix-matched standards to compensate for their influence.
- Verify Instrument Calibration:Regularly calibrate analytical instruments using certified reference materials to ensure accurate measurements.
- Train and Monitor Analysts:Provide adequate training to analysts and monitor their performance to minimize variability in recovery results.
Table of Typical Recovery Percentages
The following table provides a general overview of typical recovery percentages for various analytes and matrices. However, it’s important to note that actual recovery values may vary depending on the specific method and conditions used.
| Analyte | Matrix | Typical Recovery (%) |
|---|---|---|
| Benzene | Water | 95-105 |
| Lead | Soil | 80-120 |
| Caffeine | Coffee | 90-110 |
| Ibuprofen | Plasma | 70-130 |
| Vitamin C | Orange Juice | 98-102 |
Epilogue
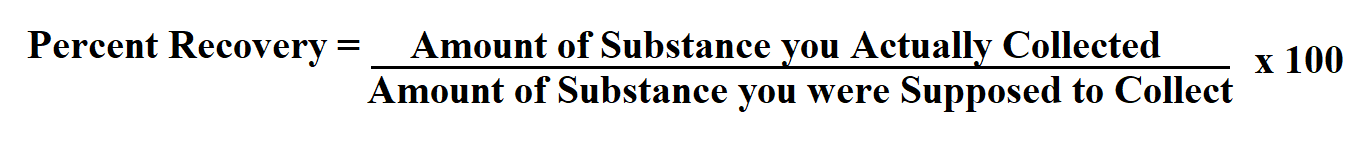
By understanding the principles and techniques discussed in this guide, you can effectively calculate percent recovery, optimize your analytical procedures, and ensure the reliability of your results.
